Assembling global maps of cellular function through integrative analysis of physical and genetic networks. Comprehensive cluster analysis with Transitivity Clustering. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Integration of biological networks and gene expression data using Cytoscape. Biana: a software framework for compiling biological interactions and analyzing networks. Garcia-Garcia, J., Guney, E., Aragues, R., Planas-Iglesias, J. Graph-based analysis and visualization of experimental results with ONDEX. VisANT: data-integrating visual framework for biological networks and modules. Pajek-program for large network analysis. 
QuACN: an R package for analyzing complex biological networks quantitatively. Mueller, L.A.J., Kugler, K.G., Dander, A., Graber, A. Node centrality in weighted networks: generalizing degree and shortest paths. statnet: software tools for the representation, visualization, analysis and simulation of network data. Handcock, M.S., Hunter, D.R., Butts, C.T., Goodreau, S.M. The igraph software package for complex network research. Proceedings of the 7th Python in Science Conference 11–15 (2008).Ĭsárdi, G. Exploring network structure, dynamics, and function using NetworkX. LEDA: A Platform for Combinatorial and Geometric Computing (Cambridge University Press, 1999). The JUNG (Java Universal Network/Graph) Framework. Tools for visually exploring biological networks. A survey of visualization tools for biological network analysis. Getting connected: analysis and principles of biological networks. Protein-protein interactions: making sense of networks via graph-theoretic modeling. Using graph theory to analyze biological networks. Analysis of Biological Networks (John Wiley & Sons, 2008). Network analysis tools: from biological networks to clusters and pathways. Biological impacts and context of network theory. Molecular basis of telaprevir resistance due to V36 and T54 mutations in the NS3-4A protease of the hepatitis C virus. Intra and inter-molecular communications through protein structure network. Proteins as networks: usefulness of graph theory in protein science. Creative elements: network-based predictions of active centres in proteins and cellular and social networks. 
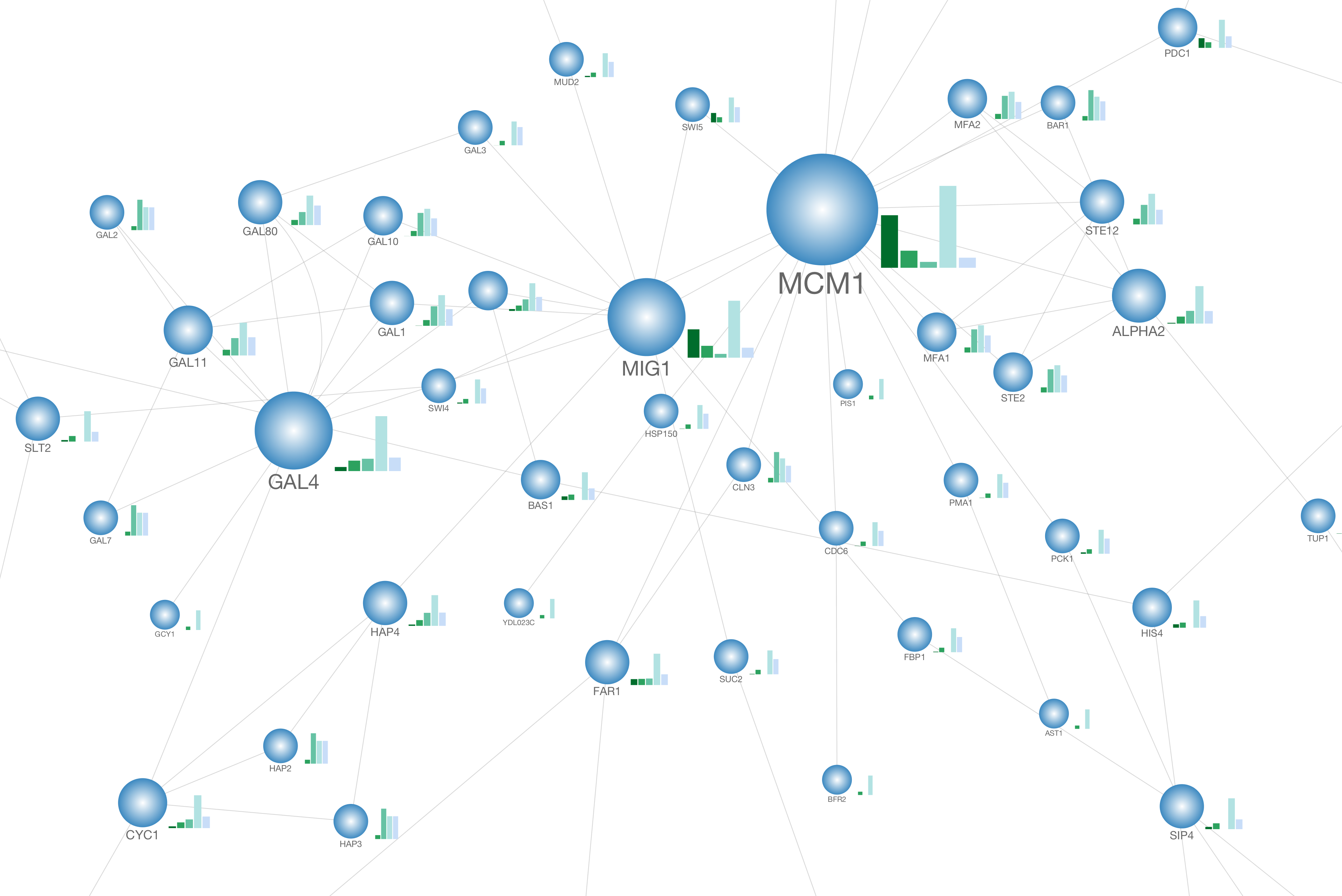
Discovering functions and revealing mechanisms at molecular level from biological networks. Evolution of biomolecular networks: lessons from metabolic and protein interactions. Toward the dynamic interactome: it's about time. in Modern Genome Annotation: the Biosapiens Network 353–410 (Springer-Verlag, 2008). Protein-protein interactions: analysis and prediction. Integrating physical and genetic maps: from genomes to interaction networks. Network medicine: a network-based approach to human disease. degree or clustering index).Barabasi, A.L., Gulbahce, N. Finally, to enrich the visual exploration, it is possible to visually render local topological properties of the network (e.g. The plugin also allows the extraction of parts of the network that contain a selected subset of reactions. the organism or perturbation under study), we propose facilities to edit/select putative biochemical transformations. Since the definition of this list is closely related to experimentation (i.e. Inference requires a list of potential biochemical transformations. Here we present a new plugin for Cytoscape dedicated to the inference and visualization of high-resolution metabolomic networks. There is currently no available software that allows inference and visualization of such high-resolution metabolomic networks directly from raw data. To analyze, explore and interpret these two kinds of relations, powerful visualization tools are required. The combination of these two inference methods generates networks containing hundreds of nodes (metabolites) and hundreds of predicted edges (biochemical reactions and/or high correlations). Moreover, perturbation studies allow the use of correlation analysis to infer/confirm links between metabolites that correlate across various conditions. Such high-resolution data has also been used to predict ab initio biochemical interactions between metabolites. Recently, ultra high-resolution mass spectrometry (FTICR-MS or Orbitrap) has been successfully used in metabolomic studies. Various spectrometric technologies are capable of identifying thousands of metabolites. Metabolomics aims at the identification and quantification of all metabolites that are present in a cell, tissue or biofluid at a given moment and under particular conditions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
